Last updated: February 28, 2012
NIH researchers identify genetic cause of anemia disorder
NIH researchers identify genetic cause of anemia disorder
DNA barrier element implicated in human disease for the first time
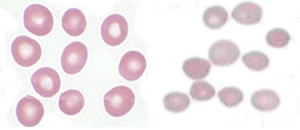 |
HS affects 1 in 2,000 individuals of Northern European ancestry and is characterized by fatigue, anemia, enlargement of the spleen and sometimes jaundice. In this study, the researchers investigated a specific mutation in the non-coding region of the ankyrin-1 (ANK1) gene, the gene most commonly mutated in HS, and sought to determine whether the mutation was responsible for the ankyrin deficiency in patients with the disorder.
"Our study provides novel understanding of barrier insulator structure and function along with the particular role of this mechanism in a human disorder," said senior author David Bodine, Ph.D., NHGRI senior investigator and chief of the Genetics and Molecular Biology Branch. "Barrier insulators are not well understood, but what we are learning is fundamental to gene regulation."
Barrier insulators are stretches of DNA that reside outside the coding region of the genome and their associate binding proteins. Unlike genes, they do not code for human proteins or carry hereditary traits, but are among the complement of DNA elements that play a role in turning genes on and off. Barrier insulators create a barrier to help keep genes 'on' in the appropriate cell type.
People contain a vast number of cell types, all differing in their structure and function. All cell nuclei within an individual contain the same set of genes, some of which may or may not be switched on by regulatory elements also written in DNA code. These include enhancers, silencers and barrier insulators to name but a few. The genes that are turned on, or expressed, in the early phase of red blood cells (fully developed red blood cells do not contain nuclei) are different from those that form a skin or muscle cell.
ANK1 produces a protein necessary for the stability of the red cell membrane. The barrier insulator defect that causes HS was detected in DNA adjacent to the ANK1 promoter, the region just before the gene in its sequence that controls RNA production. Mutations in the barrier insulator lead to silencing of ANK1 and a deficiency of ankyrin protein in the cells. Ankyrin deficiency in red blood cells causes them to become spherically shaped rather than donut shaped and their cell membranes to lose the ability to mold and flex on their way through the tiny blood vessels that branch out to bring oxygen to tissues throughout the body.
The research team used a variety of approaches to demonstrate that this barrier insulator causes the disorder, including studies that showed a loss of barrier-associated proteins at the mutant ankyrin promoter and the use of transgenic mice to demonstrate that the barrier region could function independently of the ANK1 itself.
"Many studies have identified noncoding regions of the genome that are associated with specific diseases, but the role of those sequences in disease has not been demonstrated," said the study's lead author Patrick Gallagher, M.D., Yale University professor of pediatrics and genetics. Bodine added, "I believe our study is the first of many studies that will show that mutations in barrier insulator elements are responsible for numerous inherited and acquired diseases, including cancers."
In addition to NHGRI, the study was supported by the National Institute of Diabetes and Digestive and Kidney Diseases and the National Heart, Lung, and Blood Institute.
Last Updated: February 28, 2012
