Policy Issues in Genomics
NHGRI is committed to driving the responsible use of genomics in society in order to advance knowledge and ensure that genomics benefits the health of all humans. To do this, we consider the ethical, legal, and social aspect of genomics research in our work, including these key issues.

Payers such as insurance companies and Medicare need systematic ways of evaluating genetic tests for reimbursement.

In 2008, the Genetic Information Nondiscrimination Act was passed into law, prohibiting discrimination by employers and health insurers.
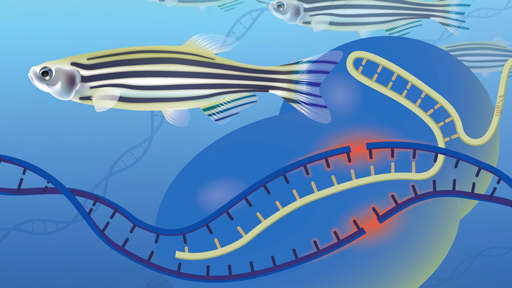
Genome editing is a method that lets scientists change the DNA of many organisms, including plants, bacteria, and animals.

NHGRI wants to ensure that all populations benefit from the advances of genomics research.

Federally-funded research with human participants must comply with regulations that protect the rights and welfare of the participants.

Informed consent shows respect for personal autonomy and is an important ethical requirement in research.

In June 2013, the Supreme Court determined that DNA in its natural form cannot be patented.

Genetic information is used by law enforcement to investigate criminal acts and exonerate those who have been convicted of crimes they did not commit.

Appropriate use of population descriptors in research is a critical scientific issue that is important for advancing genomic science and improving healthcare across human populations.

There are laws and policies that serve to protect the privacy of individuals' genomic information.

Most genetic tests today are not regulated, meaning that they go to market without any independent analysis to verify the claims of the seller.

Synthetic biology is a field of science that involves redesigning organisms for useful purposes by engineering them to have new abilities.
Last updated: February 8, 2024



