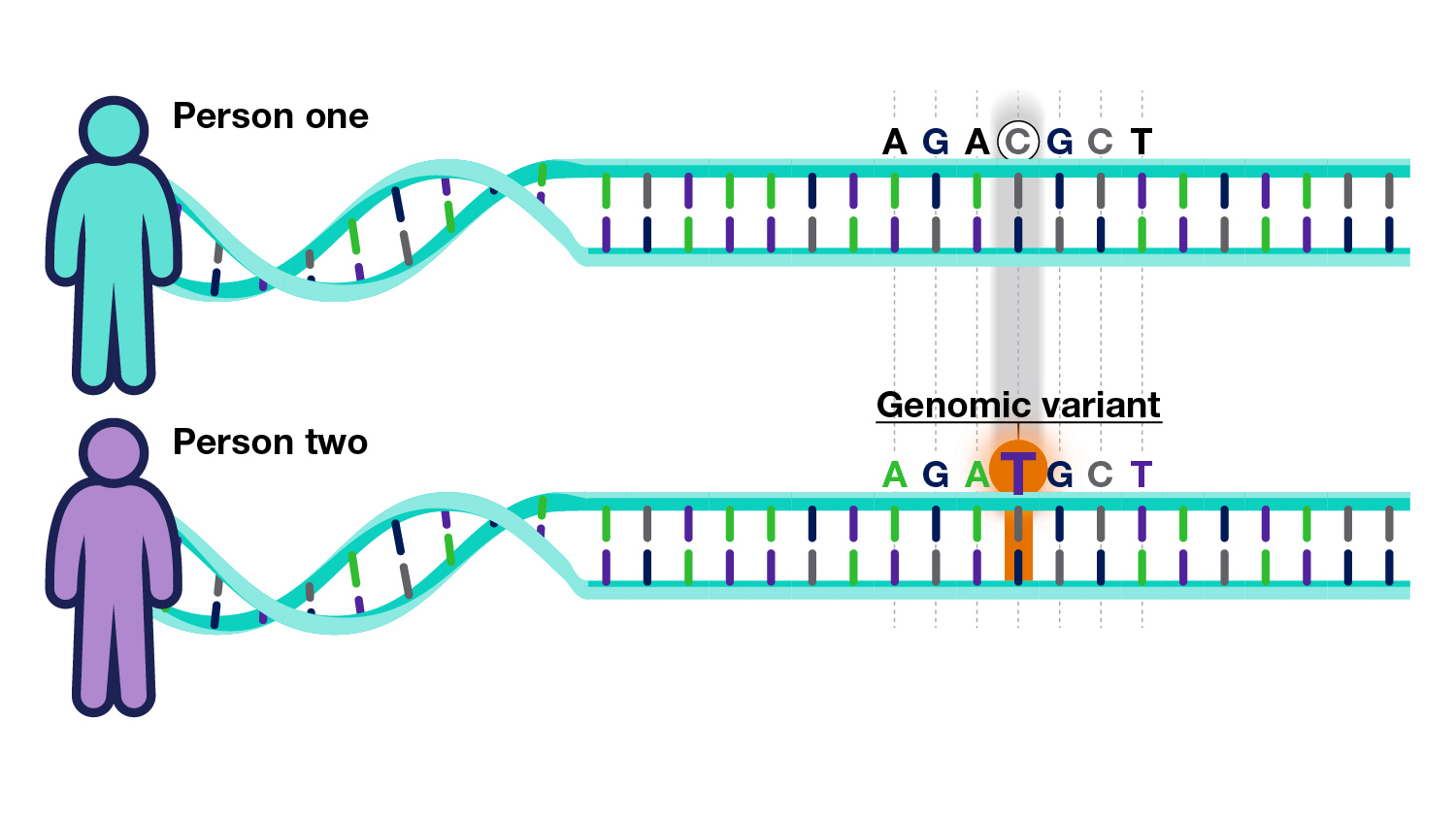
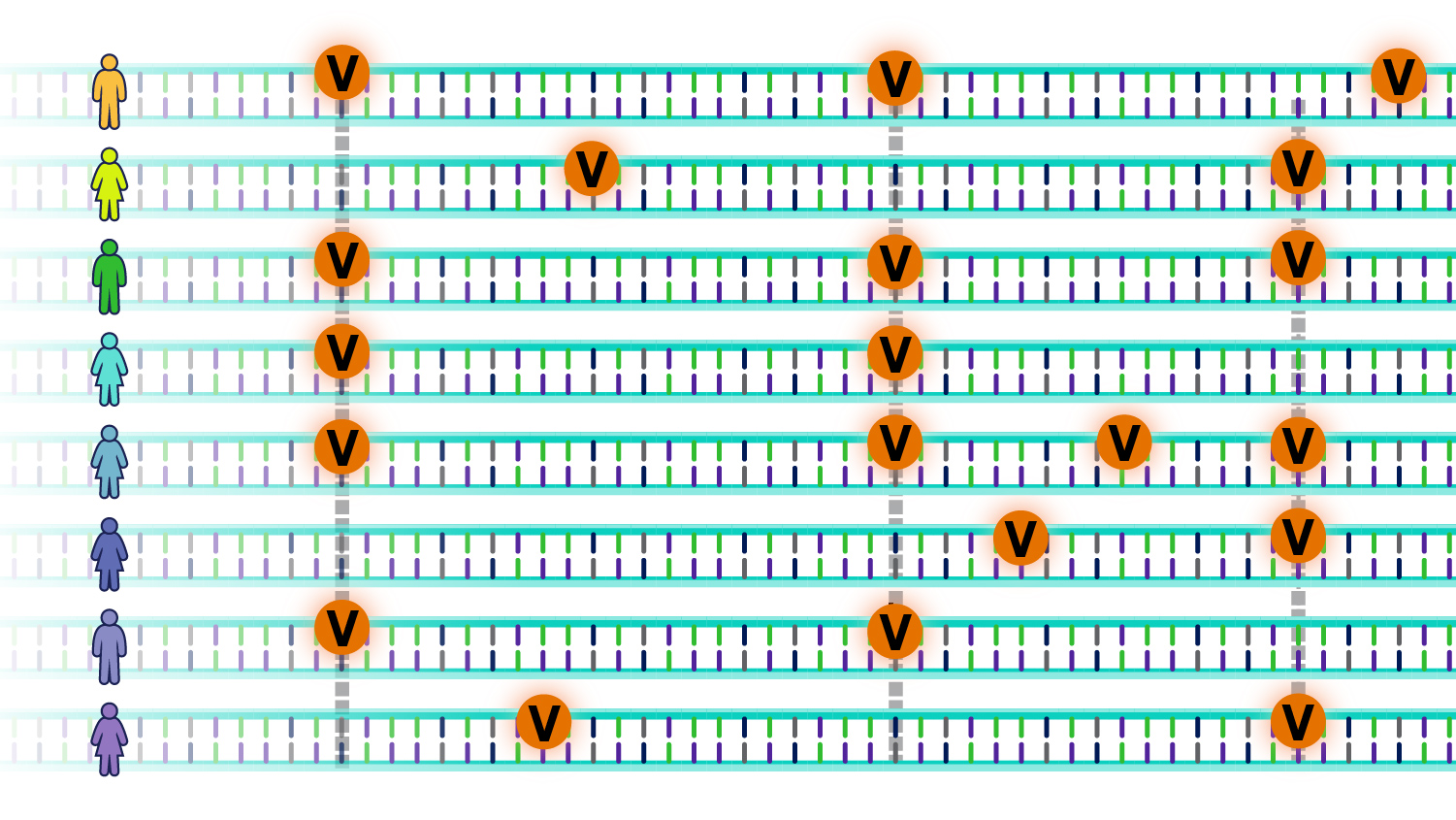
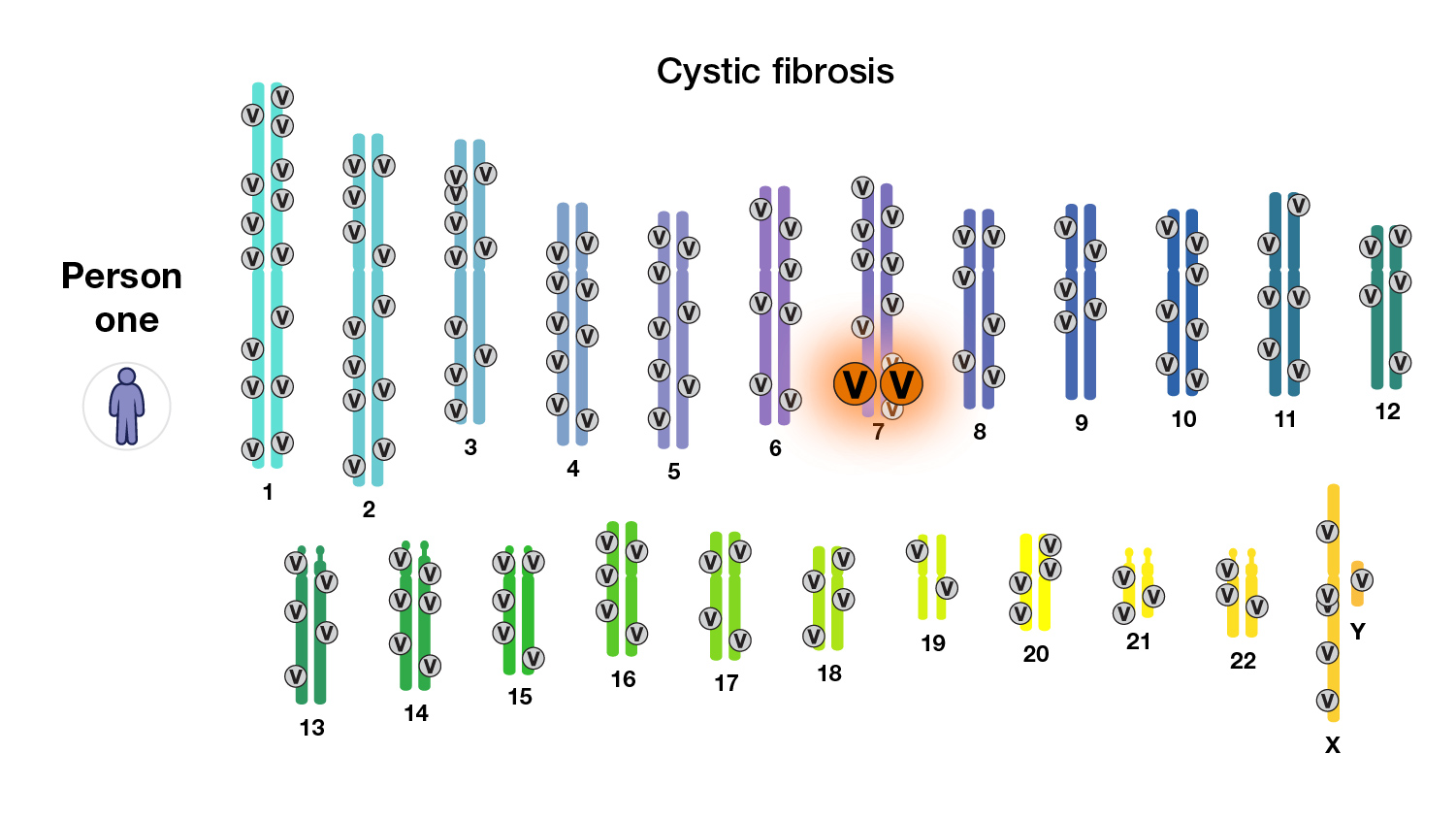
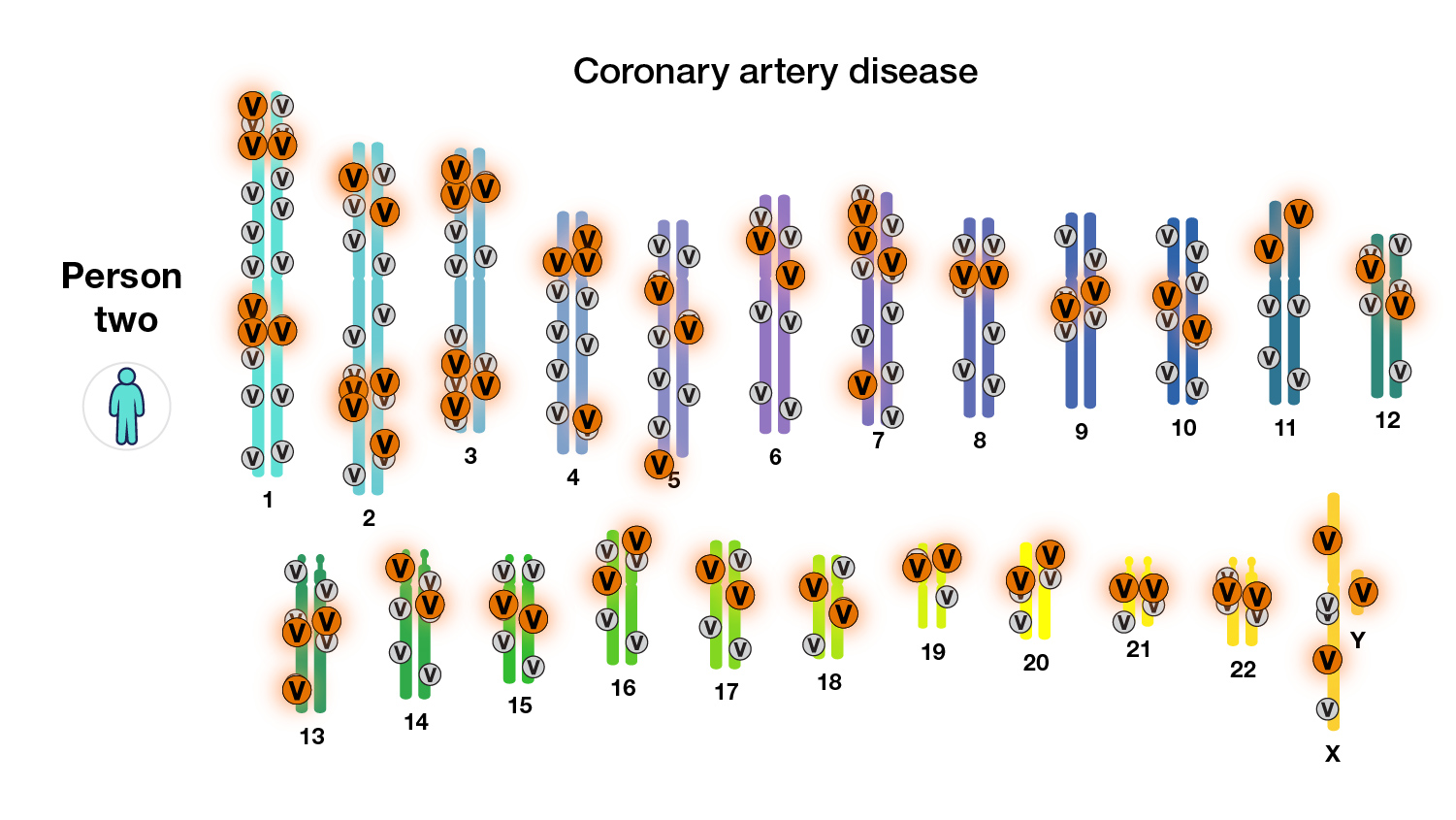
Many people have an illness, or several illnesses, that are affected by changes in either one or many of their genes, frequently coupled with environmental factors.
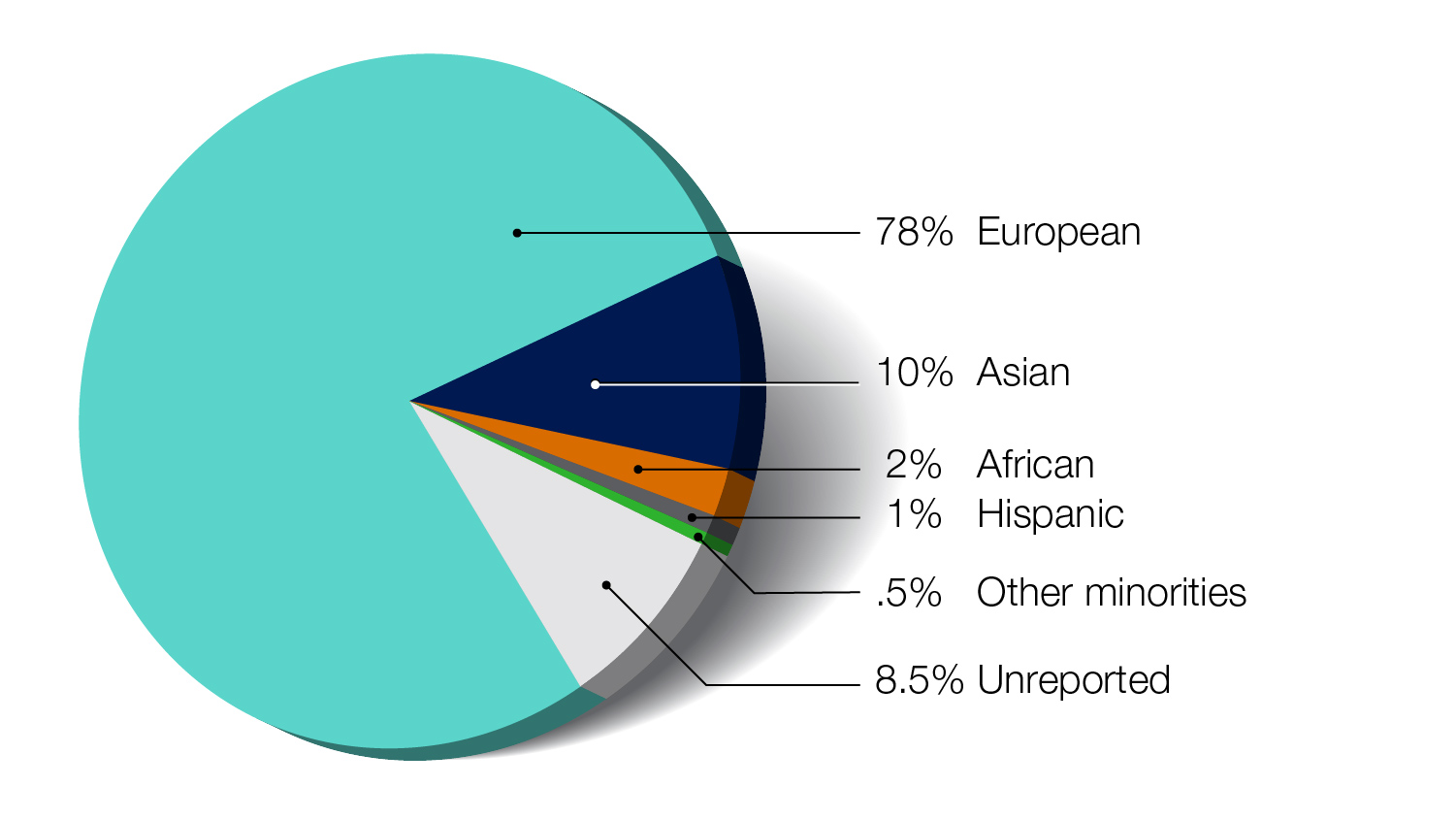
Researchers are studying these changes to understand the role that genetics plays in diseases across different populations.
A “polygenic risk score” is one way by which people can learn about their risk of developing a disease, based on the total number of changes related to the disease.
Featured video
Researchers often divide genetic diseases into two classes: those that are associated with a single gene and those that are influenced by multiple genes and environmental factors. Many diseases fall on a spectrum between these two extremes.
Last updated: August 11, 2020