Last updated: November 22, 2013
NIH Scientists Discover Crucial Control in Long-Lasting Immunity

National Institute of Allergy and Infections Diseases
www.niaid.nih.gov
National Human Genome Research Institute
www.genome.gov
NIH Scientists Discover Crucial Control in Long-Lasting Immunity
Tango Between T and B Cells Depends on Key Protein
 Bethesda, Md., Fri., Oct. 10, 2008 - National Institutes of Health (NIH) scientists have identified a protein that plays matchmaker between two key types of white blood cells, T and B cells, enabling them to interact in a way that is crucial to establishing long-lasting immunity after an infection. Their finding may also explain why some individuals who have a genetic defect that prevents them from making this protein-called SAP-suffer from lethal infections with a common virus that otherwise is rarely fatal (Epstein-Barr virus), while others with this genetic defect have problems with B-cell lymphomas.
Bethesda, Md., Fri., Oct. 10, 2008 - National Institutes of Health (NIH) scientists have identified a protein that plays matchmaker between two key types of white blood cells, T and B cells, enabling them to interact in a way that is crucial to establishing long-lasting immunity after an infection. Their finding may also explain why some individuals who have a genetic defect that prevents them from making this protein-called SAP-suffer from lethal infections with a common virus that otherwise is rarely fatal (Epstein-Barr virus), while others with this genetic defect have problems with B-cell lymphomas.
The new study was a collaboration between the laboratories of NIH scientists Ronald Germain, M.D., Ph.D., at the National Institute of Allergy and Infectious Diseases (NIAID), and Pamela Schwartzberg, M.D., Ph.D., at the National Human Genome Research Institute (NHGRI). Their findings appear in the Oct. 9 issue of Nature.
When a B cell encounters a virus or other foreign agent, the B cell engulfs the virus and parts of it, called antigens, become displayed on the surface of the B cell. These antigens signal to T cells, which have specialized receptors that can bind to antigens on the B cells. Thus coupled, the T cells deliver signals that help B cells multiply and produce antibodies that will eventually destroy the offending virus. A critical tissue structure that facilitates these events is the germinal center, which normally forms after an infection within the lymph nodes, organs in which immune cells gather to carry out their functions.
"Understanding how B and T cells interact in the lymph nodes is crucial, because the germinal centers are the sites where long-lasting immunity begins," says NIAID Director Anthony S. Fauci, M.D. "If we can unravel how the body naturally builds defenses against repeat infections, it will aid us in developing more effective vaccines."
In some individuals, B cells do not make antibodies in response to infections and immunizations. These individuals include those with a genetic disease called X-linked lymphoproliferative disease (XLP), a rare disorder affecting approximately one in one million male children. Those affected with XLP lack SAP protein and do not have germinal centers. As a result, they have an impaired immune response to certain infections, which can prove fatal.
According to Dr. Germain, "For years, researchers tried to understand what the relationship was between the absence of germinal centers and the missing SAP protein in XLP patients by studying mouse models of the human disease, but we could not identify why the absence of SAP caused these problems."
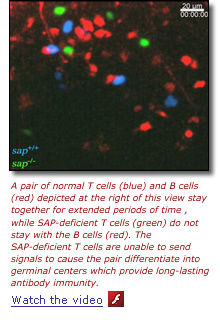
The work by Hai Qi, M.D., Ph.D., and Jennifer Cannons, Ph.D., in the laboratories of Drs. Germain and Schwartzberg, respectively, has now solved an important piece of this puzzle. Using a special microscopy technique that allows direct visualization of immune cell actions in living mice, they observed how T and B cells and another type of white blood cell, the dendritic cell, interact dynamically within mouse lymph nodes. The research team found that T cells lacking SAP do not bind strongly to B cells carrying antigens the T cells otherwise could recognize.
"The relationship between B and T cells normally is like a pair of dancers," says Dr. Qi, the lead author. "The B cell leads and the T cell follows in a long tango that eventually goes into the germinal center."
Without SAP, however, it is as if the arms of the T-cell receptor cannot firmly grasp the antigenic arms of the B cell, and thus the tango between the two immune cells does not last. The inability of SAP-negative T cells to adhere to B cells prevents B cells from receiving crucial signals they need to become antibody-secreting cells and to generate functional germinal centers.
Another striking finding was that that SAP-negative T cells can bind to dendritic cells without any problems, although dendritic cells have antigenic arms similar to those of B cells. Dr. Cannons explains, "Some cell receptors act like Velcro, allowing two different cells to stick together. We think that different cells use different receptor signals to stick together. A signal that does not affect the sticking between two cell types may well prevent another two cell types from interacting."
These findings not only help explain the basis for the immunodeficiency disease known as XLP, but also provide important new insights into the exquisitely specific communications between different types of immune cells.
According to Dr. Schwartzberg, "This work provides a great example of the power of combining mouse models of human diseases with live imaging microscopy to uncover secrets of the immune system and help discover new ways to combat disease."
"This work nicely demonstrates both the value of interdisciplinary collaboration and that exploring relatively rare genetic diseases can offer important insights into biological processes that are important to the health of all of us," says Alan E. Guttmacher, M.D., acting director, NHGRI.
NIAID conducts and supports research-at NIH, throughout the United States, and worldwide-to study the causes of infectious and immune-mediated diseases, and to develop better means of preventing, diagnosing and treating these illnesses. News releases, fact sheets and other NIAID-related materials are available on the NIAID Web site at www.niaid.nih.gov.
The National Institutes of Health (NIH) - The Nation's Medical Research Agency - includes 27 Institutes and Centers and is a component of the U. S. Department of Health and Human Services. It is the primary federal agency for conducting and supporting basic, clinical and translational medical research, and it investigates the causes, treatments and cures for both common and rare diseases. For more information about NIH and its programs, visit www.nih.gov.
To view the PDF on this page you will need Adobe Reader. ![]()
![]() To view the flash presentation on this page, you will need Adobe Flash Player installed. Download the latest version of Adobe Flash Player.
To view the flash presentation on this page, you will need Adobe Flash Player installed. Download the latest version of Adobe Flash Player.
Contacts:
Julie Wu, NIAID
301-402-1663
wujuli@niaid.nih.gov
Raymond MacDougall
NIH/NHGRI
301-402-0911
macdougallr@mail.nih.gov
