Last updated: July 15, 2013
Bioluminescent comb jellies begin to shed light on the evolution of vision
Bioluminescent comb jellies begin to shed light on the evolution of vision
By Jeannine Mjoseth
Deputy Chief, Communications and Public Liaison Branch
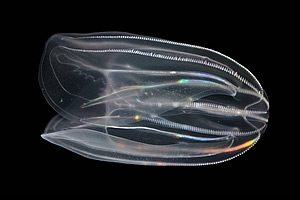
Comb jellies, also known as ctenophores, evolved more than 500 million years ago. They're among the earliest metazoans, a group comprised of all multicellular animals. At a maximum length of five inches, they have the distinction of being the largest animals to use cilia for locomotion. Ctenophores are found in all oceans of the world and have recently invaded the Black, Caspian and Baltic Seas, where they are considered pests because they consume fish larvae. The subject of this study was a comb jelly called Mnemiopsis leidyi that lives off the eastern seaboard, from Maine to Florida.
The study was performed by Dr. Andy Baxevanis and his group in the Genome Technology Branch of the National Human Genome Research Institute (NHGRI). They based their findings on the whole genome of Mnemiopsis leidyi, which the group recently sequenced, assembled and annotated. Dr. Baxevanis and his group will soon publish a study analyzing the organism's genome, the first bioluminescent animal and the first ctenophore species for which there is a whole genome sequence.
"Comb jellies are quite beautiful," said Christine E. Schnitzler, Ph.D., the paper's first author and a postdoctoral fellow in Dr. Baxevanis' group. "When light reflects off their cilia, it creates a rainbow of colors. But that's not the same as bioluminescence, which you can only see in the dark."
Comb jellies' ability to generate light comes from genes that produce photoproteins. Two types of chemicals involved in light production, called luciferin and luciferase, are bound together in a photoprotein. This molecule can be triggered to produce light when calcium is added to the system. Photoproteins emit flashes of very bright light for a fraction of a second. In the animal, this can translate to an impressive display of lights that run up and down the animal.
While the researchers expected to find photoproteins in comb jellies, they didn't expect to find 10 different photoproteins that were clustered into two groups in the genome. The researchers think that having multiple photoprotein genes allows the animal to produce larger amounts of photoprotein and emit more light quickly.
"These protoproteins were tandemly arrayed, meaning that they were arranged one after another," Dr. Schnitzler said. "This is fascinating for an evolutionary biologist because it means that these photoproteins somehow evolved as a group."
Researchers found that light-emitting photoproteins were located in the same cells as opsin genes, the most primitive type of light detector in animals. All animals use opsins to catch photons of light, and finding a functional opsin in a ctenophore indicates that they have been preserved throughout animal evolution.
The co-localization of the opsin genes and the photoproteins may confer an advantage to the animal: it might be a feedback mechanism that allows the comb jelly to maintain the right amount of bioluminescence or it may enhance reproductive or defensive capabilities, Dr. Schnitzler suggested.
Opsins are also located near the apical sensory organ, which is opposite the mouth. An apical sensory organ contains large sensory cilia that help it maintain proper vertical orientation or change swimming direction.
Dr. Baxevanis sees a broader context for using comparative genomic approaches focused on the early animals such as comb jellies.
"This study provides a good example of what we can learn from a non-traditional model organism," Dr. Baxevanis said. "To get a handle on the evolution of vision, we first need to understand the evolution of protein families like the photoproteins and the opsins. These organisms may also help us figure out at what point in time a particular disease-associated gene emerged."