Biological Pathways Fact Sheet
A biological pathway is a series of actions among molecules in a cell that leads to a certain product or a change in the cell. It can trigger the assembly of new molecules, such as a fat or protein, turn genes on and off, or spur a cell to move.
How do biological pathways work?
For your body to develop properly and stay healthy, many things must work together at many different levels - from organs to cells to genes.
From both inside and outside the body, cells are constantly receiving chemical cues prompted by such things as injury, infection, stress or even the presence or lack of food. To react and adjust to these cues, cells send and receive signals through biological pathways. The molecules that make up biological pathways interact with signals, as well as with each other, to carry out their designated tasks.
Biological pathways can act over short or long distances. For example, some cells send signals to nearby cells to repair localized damage, such as a scratch on a knee. Other cells produce substances, such as hormones, that travel through the blood to distant target cells.
These biological pathways control a person's response to the world. For example, some pathways subtly affect how the body processes drugs, while others play a major role in how a fertilized egg develops into a baby. Other pathways maintain balance while a person is walking, control how and when the pupil in the eye opens or closes in response to light, and affect the skin's reaction to changing temperature.
Biological pathways do not always work properly. When something goes wrong in a pathway, the result can be a disease such as cancer or diabetes.
-
How do biological pathways work?
For your body to develop properly and stay healthy, many things must work together at many different levels - from organs to cells to genes.
From both inside and outside the body, cells are constantly receiving chemical cues prompted by such things as injury, infection, stress or even the presence or lack of food. To react and adjust to these cues, cells send and receive signals through biological pathways. The molecules that make up biological pathways interact with signals, as well as with each other, to carry out their designated tasks.
Biological pathways can act over short or long distances. For example, some cells send signals to nearby cells to repair localized damage, such as a scratch on a knee. Other cells produce substances, such as hormones, that travel through the blood to distant target cells.
These biological pathways control a person's response to the world. For example, some pathways subtly affect how the body processes drugs, while others play a major role in how a fertilized egg develops into a baby. Other pathways maintain balance while a person is walking, control how and when the pupil in the eye opens or closes in response to light, and affect the skin's reaction to changing temperature.
Biological pathways do not always work properly. When something goes wrong in a pathway, the result can be a disease such as cancer or diabetes.
What are some types of biological pathways?
There are many types of biological pathways. Among the most well-known are pathways involved in metabolism, in the regulation of genes and in the transmission of signals.
Metabolic pathways make possible the chemical reactions that occur in our bodies. An example of a metabolic pathway is the process by which cells break down food into energy molecules that can be stored for later use. Other metabolic pathways actually help to build molecules.
Gene-regulation pathways turn genes on and off. Such action is vital because genes provide the recipe by which cells produce proteins, which are the key components needed to carry out nearly every task in our bodies. Proteins make up our muscles and organs, help our bodies move and defend us against germs.
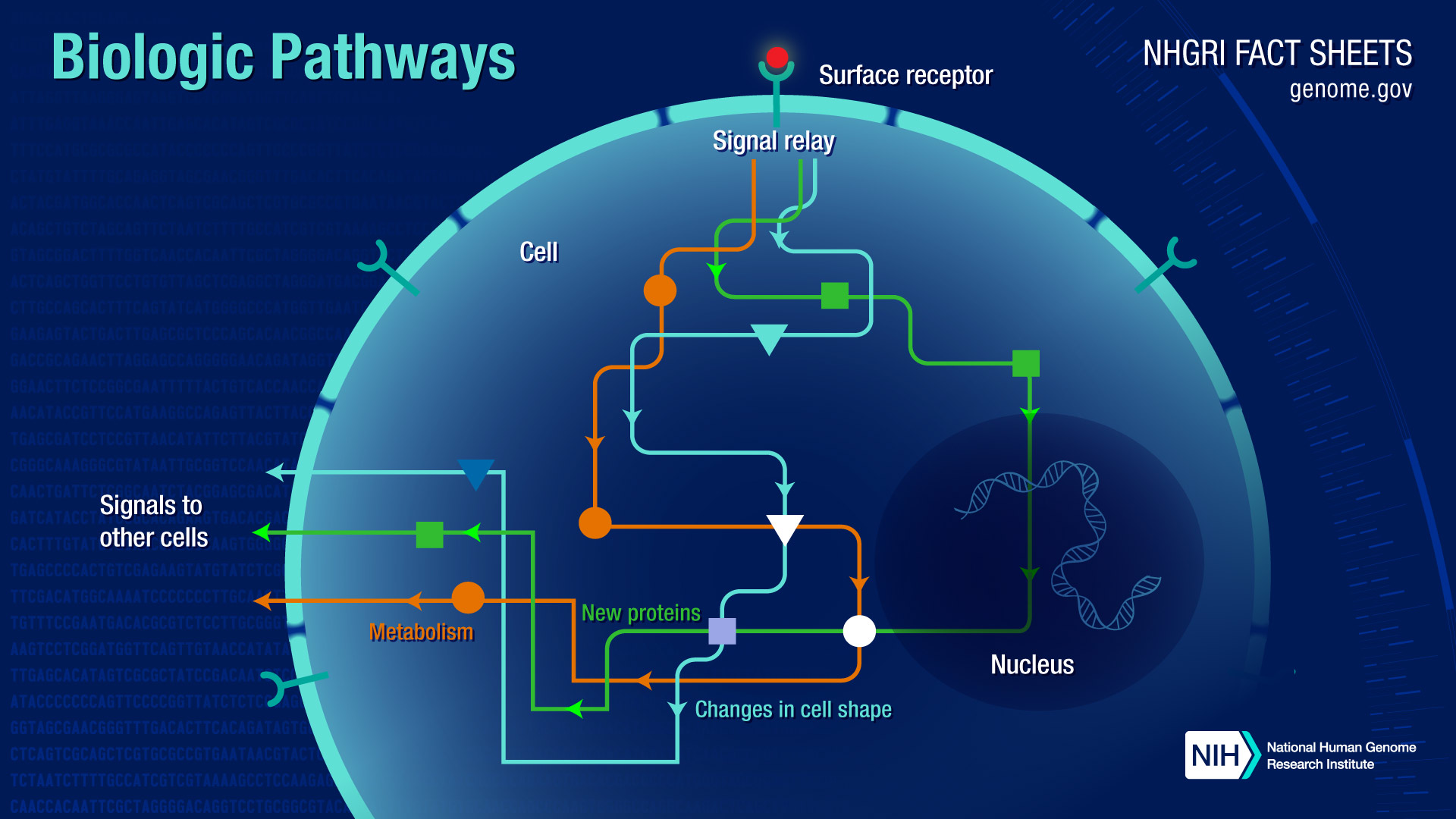
Signal transduction pathways move a signal from a cell's exterior to its interior. Different cells are able to receive specific signals through structures on their surface called receptors. After interacting with these receptors, the signal travels into the cell, where its message is transmitted by specialized proteins that trigger a specific reaction in the cell. For example, a chemical signal from outside the cell might direct the cell to produce a particular protein inside the cell. In turn, that protein may be a signal that prompts the cell to move.
-
What are some types of biological pathways?
There are many types of biological pathways. Among the most well-known are pathways involved in metabolism, in the regulation of genes and in the transmission of signals.
Metabolic pathways make possible the chemical reactions that occur in our bodies. An example of a metabolic pathway is the process by which cells break down food into energy molecules that can be stored for later use. Other metabolic pathways actually help to build molecules.
Gene-regulation pathways turn genes on and off. Such action is vital because genes provide the recipe by which cells produce proteins, which are the key components needed to carry out nearly every task in our bodies. Proteins make up our muscles and organs, help our bodies move and defend us against germs.
Signal transduction pathways move a signal from a cell's exterior to its interior. Different cells are able to receive specific signals through structures on their surface called receptors. After interacting with these receptors, the signal travels into the cell, where its message is transmitted by specialized proteins that trigger a specific reaction in the cell. For example, a chemical signal from outside the cell might direct the cell to produce a particular protein inside the cell. In turn, that protein may be a signal that prompts the cell to move.

What is a biological network?
Researchers are learning that biological pathways are far more complicated than once thought. Most pathways do not start at point A and end at point B. In fact, many pathways have no real boundaries, and pathways often work together to accomplish tasks. When multiple biological pathways interact with each other, they form a biological network.
-
What is a biological network?
Researchers are learning that biological pathways are far more complicated than once thought. Most pathways do not start at point A and end at point B. In fact, many pathways have no real boundaries, and pathways often work together to accomplish tasks. When multiple biological pathways interact with each other, they form a biological network.
How do researchers find biological pathways?
Researchers have discovered many important biological pathways through laboratory studies of cultured cells, bacteria, fruit flies, mice and other organisms. Many of the pathways identified in these model systems are the same as, or are similar to, counterparts in humans.
Still, many biological pathways remain to be discovered. It will take years of research to identify and understand the complex connections among all the molecules in all biological pathways, as well as to understand how these pathways work together.
-
How do researchers find biological pathways?
Researchers have discovered many important biological pathways through laboratory studies of cultured cells, bacteria, fruit flies, mice and other organisms. Many of the pathways identified in these model systems are the same as, or are similar to, counterparts in humans.
Still, many biological pathways remain to be discovered. It will take years of research to identify and understand the complex connections among all the molecules in all biological pathways, as well as to understand how these pathways work together.
What can biological pathways tell us about disease?
Researchers are able to learn a lot about human disease from studying biological pathways. Identifying what genes, proteins and other molecules are involved in a biological pathway can provide clues about what goes wrong when a disease strikes.
For example, researchers may compare certain biological pathways in a healthy person to the same pathways in a person with a disease to discover the roots of the disorder. Keep in mind that problems in any number of steps along a biological pathway can often lead to the same disease.
-
What can biological pathways tell us about disease?
Researchers are able to learn a lot about human disease from studying biological pathways. Identifying what genes, proteins and other molecules are involved in a biological pathway can provide clues about what goes wrong when a disease strikes.
For example, researchers may compare certain biological pathways in a healthy person to the same pathways in a person with a disease to discover the roots of the disorder. Keep in mind that problems in any number of steps along a biological pathway can often lead to the same disease.
How can biological pathway information improve health?
Finding out what pathway is involved in a disease - and identifying which step of the pathway is affected in each patient - may lead to more personalized strategies for diagnosing, treating and preventing disease.
Researchers currently are using information about biological pathways to develop new and more effective drugs. It likely will take some time before we routinely see specifically designed drugs that are based on information about biological pathways. However, doctors are already beginning to use pathway information to choose and combine existing drugs more effectively.
-
How can biological pathway information improve health?
Finding out what pathway is involved in a disease - and identifying which step of the pathway is affected in each patient - may lead to more personalized strategies for diagnosing, treating and preventing disease.
Researchers currently are using information about biological pathways to develop new and more effective drugs. It likely will take some time before we routinely see specifically designed drugs that are based on information about biological pathways. However, doctors are already beginning to use pathway information to choose and combine existing drugs more effectively.
Why are cancer researchers excited about biological pathways?
Until recently, many researchers hoped that most forms of cancer were driven by single genetic mutations and could be treated by drugs that target those specific mutations. Much of that hope was based on the success of imatinib (Gleevec), a drug that was specifically designed to treat a blood cancer called chronic myeloid leukemia (CML). CML occurs because of a single genetic glitch that leads to the production of a defective protein that spurs uncontrolled cell growth. Gleevec binds to that protein, stopping its activity and producing dramatic results in many CML patients.
Unfortunately, the one-target, one-drug approach has not held up for most other types of cancer. Recent projects that deciphered the genomes of cancer cells have found an array of different genetic mutations that can lead to the same cancer in different patients.
Thus, instead of attempting to discover ways to attack one well-defined genetic enemy, researchers now face the prospect of fighting many enemies.
Fortunately, this complex view can be simplified by looking at which biological pathways are disrupted by the genetic mutations. With further research on biological pathways and the genetic profiles of particular tumors, drug developers might be able to focus their attention on just two or three pathways. Patients could then receive the one or two drugs most likely to repair the pathways affected in their particular tumors.
-
Why are cancer researchers excited about biological pathways?
Until recently, many researchers hoped that most forms of cancer were driven by single genetic mutations and could be treated by drugs that target those specific mutations. Much of that hope was based on the success of imatinib (Gleevec), a drug that was specifically designed to treat a blood cancer called chronic myeloid leukemia (CML). CML occurs because of a single genetic glitch that leads to the production of a defective protein that spurs uncontrolled cell growth. Gleevec binds to that protein, stopping its activity and producing dramatic results in many CML patients.
Unfortunately, the one-target, one-drug approach has not held up for most other types of cancer. Recent projects that deciphered the genomes of cancer cells have found an array of different genetic mutations that can lead to the same cancer in different patients.
Thus, instead of attempting to discover ways to attack one well-defined genetic enemy, researchers now face the prospect of fighting many enemies.
Fortunately, this complex view can be simplified by looking at which biological pathways are disrupted by the genetic mutations. With further research on biological pathways and the genetic profiles of particular tumors, drug developers might be able to focus their attention on just two or three pathways. Patients could then receive the one or two drugs most likely to repair the pathways affected in their particular tumors.
Last updated: August 15, 2020
