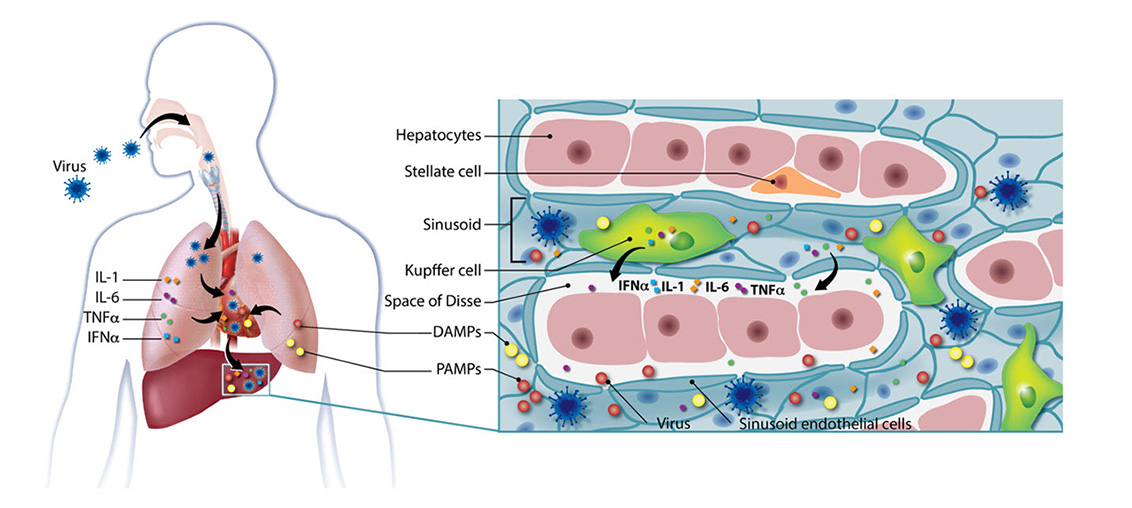
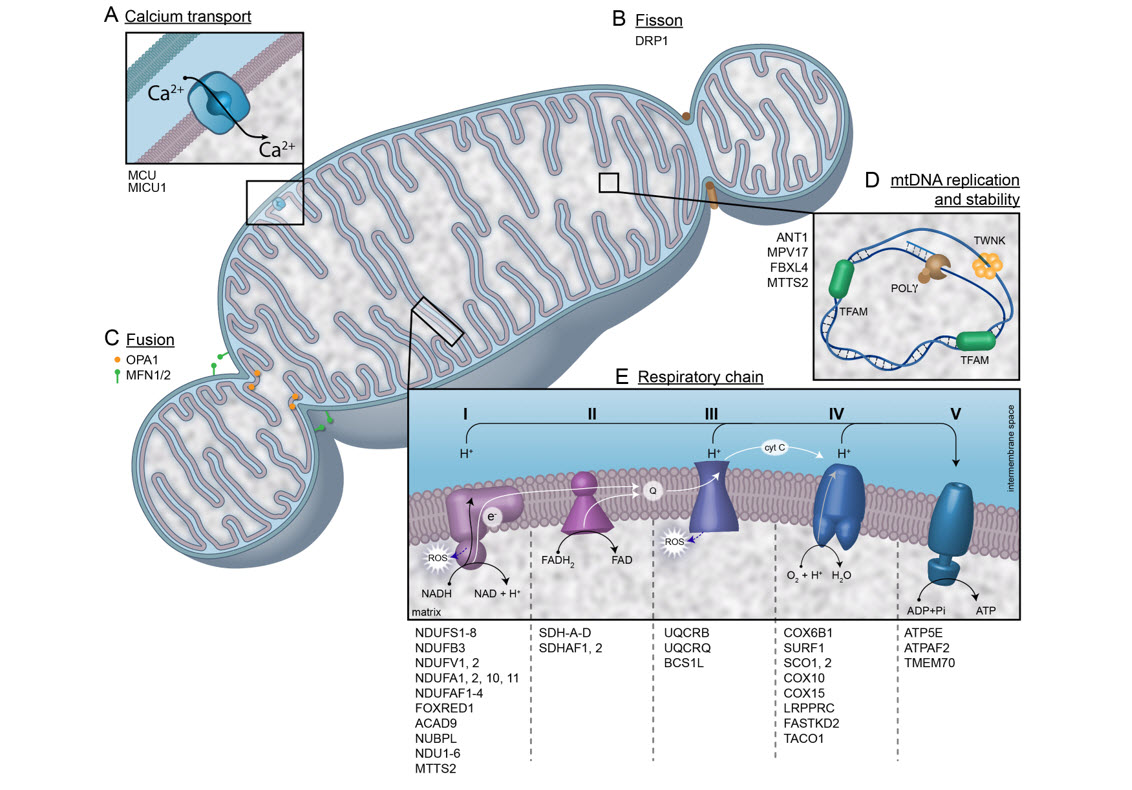
Peter McGuire, M.S., M.B.B.Ch.
Investigator
Metabolic Infection and Immunity Section
Head
Metabolism, Infection and Immunity Section
Education
B.A. Villanova University, 1993
M.S. New York Medical College, 1998
M.B.B.Ch. Royal College of Surgeons in Ireland, 2003
Metabolism, Infection and Immunity Section Staff

Tatiana Tarasenko, M.D., Ph.D.
- Staff Scientist
- Metabolism, Infection and Immunity Section

Eliza Gordon-Lipkin, M.D.
- Staff Clinician
- Metabolic Medicine Branch

Amanda Fuchs, Ph.D.
- Postdoctoral Fellow
- Metabolism, Infection and Immunity Section

Bharati Singh, PhD
- Postdoctoral Fellow
- Metabolism, Infection and Immunity Section

Shannon Kruk, R.N., B.S.
- Research Nurse Specialist
- Metabolism, Infection and Immunity Section

Melissa Schlein
- Postbaccalaureate Fellow
- Metabolism, Infection and Immunity Section

Madeleine Hesselgesser
- Postbaccalaureate Fellow
- Metabolic Infection and Immunity Section
Last updated: October 8, 2024