Cynthia Tifft, M.D., Ph.D.
Office Of The Clinical Director
Medical Genetics Branch
Glycosphingolipid and Glycoprotein Disorders Unit
B.A. University of California, San Diego
M.S. Rutgers University
Ph.D. University of Texas, Houston
M.D. University of Texas, Houston
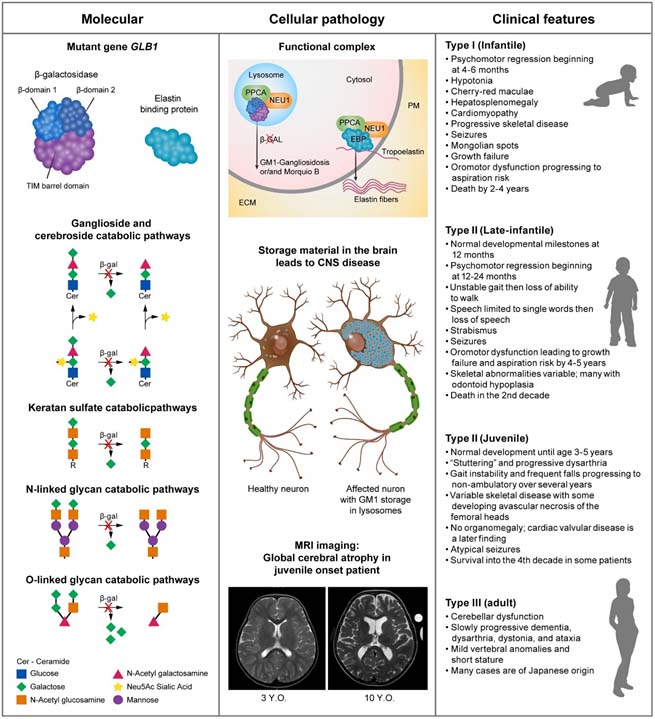
GM1 Gangliosidosis
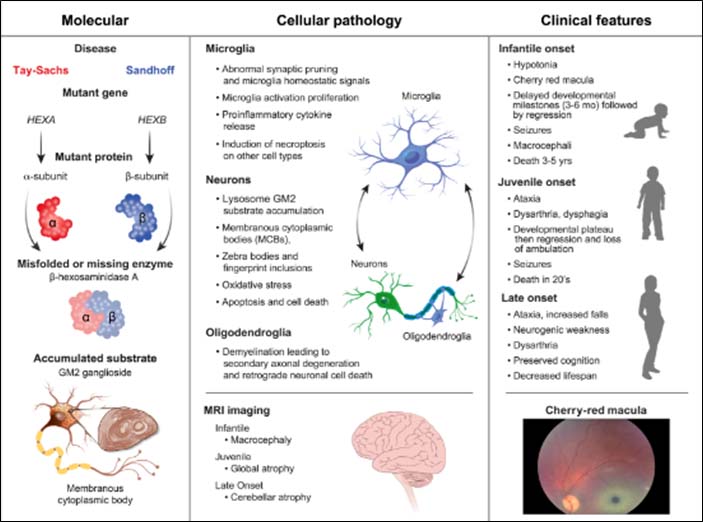
Toro C, Zainab M, Tifft CJ. The GM2 gangliosidoses: Unlocking the mysteries of pathogenesis and treatment. Neurosci Lett. 2021 Nov 1;764:136195. doi: 10.1016/j.neulet.2021.136195. Epub 2021 Aug 25. PMID: 34450229; PMCID: PMC8572160.
GM2 Gangliosidosis
Nicoli ER, Annunziata I, d'Azzo A, Platt FM, Tifft CJ, Stepien KM. GM1 Gangliosidosis-A Mini-Review. Front Genet. 2021 Sep 3;12:734878. doi: 10.3389/fgene.2021.734878. PMID: 34539759; PMCID: PMC8446533.
Glycosphingolipid Disorders and Glycoprotein Disorders Unit

- Research Nurse Coordinator
- Office of the Clinical Director

- Pediatric Nurse Practitioner
- NIH Undiagnosed Diseases Program

- Postbaccalaureate Fellow
- Glycosphingolipid and Glycoprotein Disorders Unit

- Postbaccalaureate Fellow
- Glycosphingolipid Disorders and Glycoprotein Disorders Unit
Last updated: January 12, 2025