Sickle Cell Disease
Definition
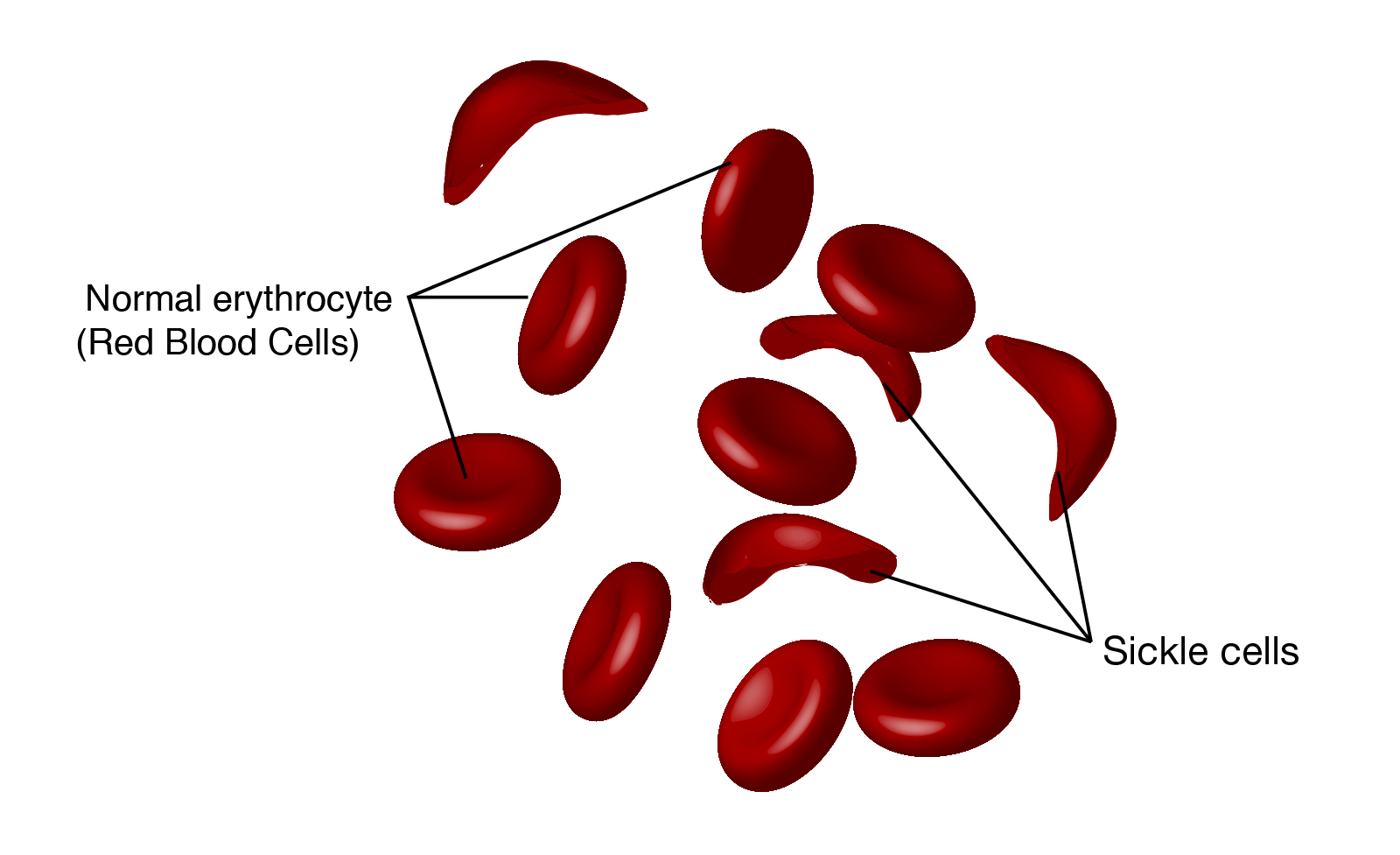
Sickle cell disease is a hereditary disease seen most often among people of African ancestry. Caused by mutations in one of the genes that encode the hemoglobin protein, the disease is inherited as an autosomal recessive trait. The mutation causes the red blood cells to take on an unusual sickle shape. Individuals affected by sickle cell disease are chronically anemic and experience significant damage to their heart, lungs, and kidneys.
Narration
Sickle cell disease is the first human inherited disease that was understood at the protein and the DNA level. Sickle cell is a disease that's primarily seen in people of African descent. In studying the genomics of people from Africa, it's very clear now that three different times during the history of the human race the mutation in the beta-globin gene that changes an amino acid at the sixth position of the protein, the same mutation has happened. And this has been expanded throughout history in Africa in three different groups and then migrated all over the world. Now, the mutation does not really affect the ability of beta-globin to participate with alpha-globin and make hemoglobin, and it doesn't affect the ability to carry oxygen. In matter of fact, people who inherit a hemoglobin S gene from their mother and a hemoglobin S gene from their father, so their homozygous for hemoglobin S, have perfectly normal oxygen-carrying capacity of hemoglobin. The problems happen when the red cell containing hemoglobin S gets into the muscles or into your brain and discharges the oxygen. And what happens is that hemoglobin S has a tendency when the oxygen goes off to stick to each other, and they form these polymers. Now, when they get oxygen back on, they go back into solution. The difference between hemoglobin S and wild-type hemoglobin is that the wild-type hemoglobin is soluble when it's got oxygen, and it's also soluble when it doesn't have oxygen. Now, if everything is working well, these polymers in people with sickle cell disease don't get very big. But that can be worsened when you have extreme oxygen stress, and that's what causes the characteristic sickle cells. The polymers get very, very long, and they stretch the cell out of shape. Now your spleen is very good at raking these cells out of the peripheral blood as they come through. And that's what causes the anemia, and the old name for sickle cell was sickle cell anemia. However, though, short polymers are very, very dangerous. If you think of a red blood cell going through your veins and arteries and capillaries as a water balloon, you'll see how it can squish itself down into a long cylinder to get through a capillary and flatten out like a pancake to get through areas in the spleen that kind of rake out the bad-shape red blood cells, and then to form right back to a regular water balloon shape to go into the arteries and veins. But if you have a red blood cell full of these small hemoglobin S polymers, it's like having a water balloon full of ice chips, and as that goes through the capillaries and the small veins, it tears up the lining of these things, just as if you had a cut, and it activates your clotting response and micro-clots will form. And sometimes these get to be bigger and bigger clots, and so the real lethality in sickle cell disease is not from the anemia, it's from this vascular disease. And so it's very unfortunate that about one-third of people who are homozygous for hemoglobin S will have one or more strokes before they're 10 years old. And in those fortunate few that are able to get to be adolescents, these clots can build up in the lungs and give a very severe disease called acute chest syndrome, which is basically emphysema or lung destruction from these obstructions. About one-third of the patients will live healthily to adulthood, but they have many problems with iron-overloaded organs, and their life span is significantly shorter. And it is simply amazing to me that this all comes from one very small change, changes in amino acid, that really isn't having any effect on the normal function of the protein, but has big effects on all of the other systems in the body that the red cells pass through.