Lyonization
Definition
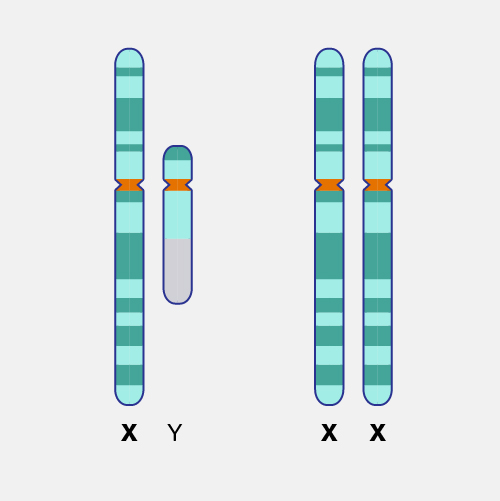
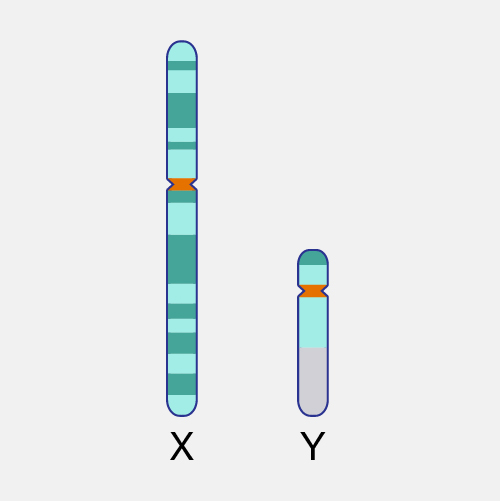
Lyonization (also called X-inactivation) refers to the normal phenomenon in which one of the two X chromosomes in every cell of a female individual is inactivated during embryonic development. This inactivation prevents females from having twice as many X chromosome gene products as males, who possess only a single copy of the X chromosome. Lyonization is named after Mary F. Lyon, the British geneticist who discovered the phenomenon.
Narration
Lyonization. Lyonization can be very important in clinical genetics. For example, imagine a family with an X-linked condition. In this family, many people might have the genetic change that can cause the condition. Part of the explanation about why some women in the family could be more severely affected than other women in the family can be because of this process of lyonization. That is, how the condition affects a person can depend on the pattern of X-inactivation or lyonization.