Frameshift Mutation
Definition
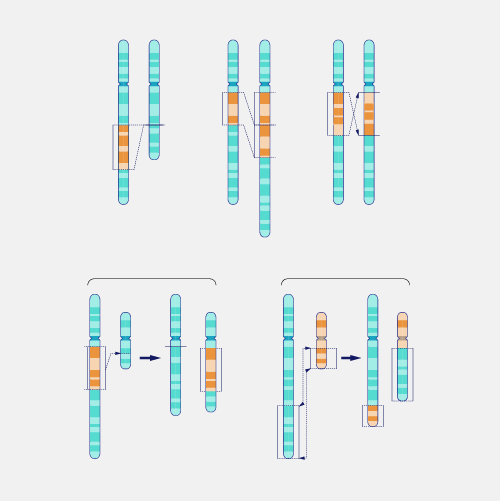
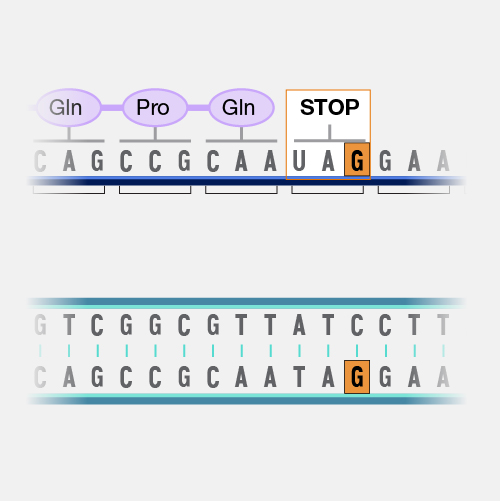
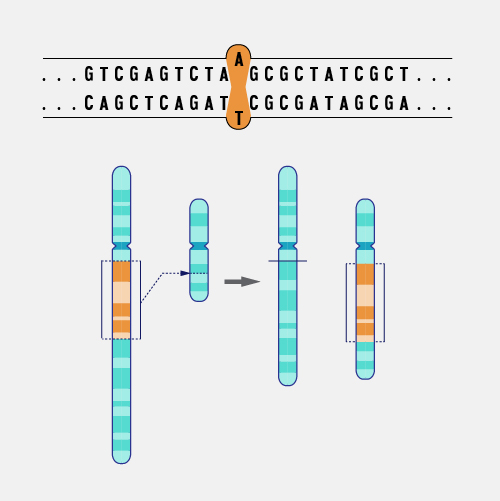
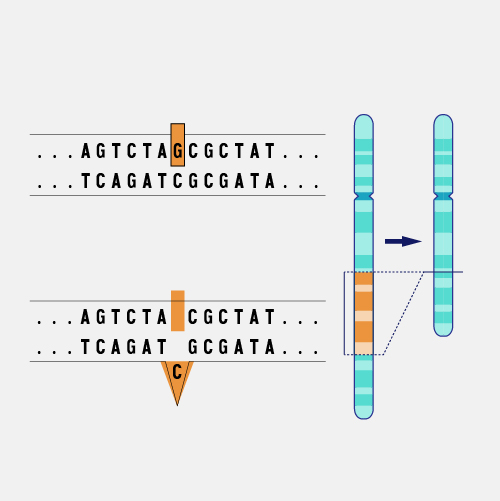
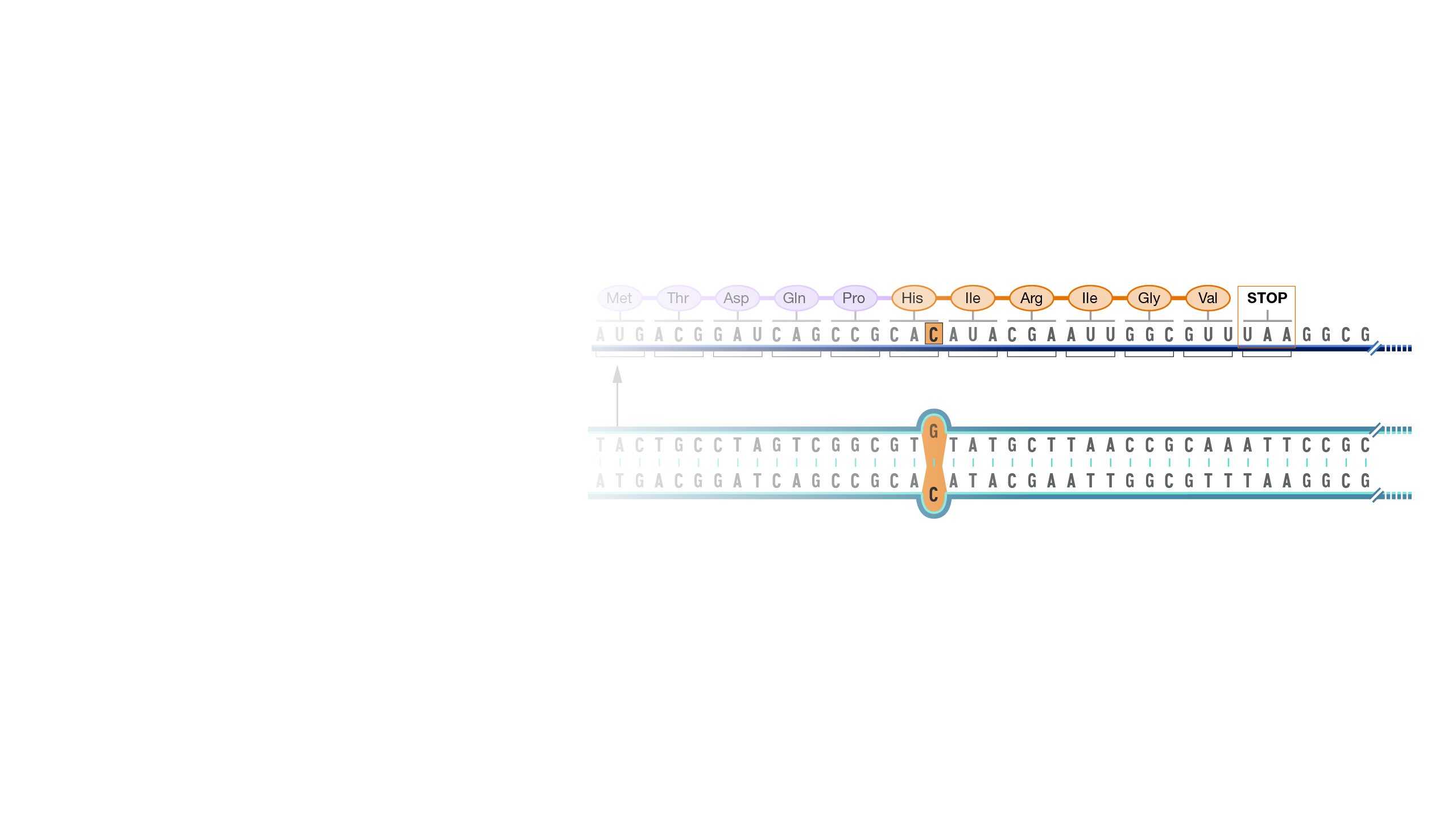
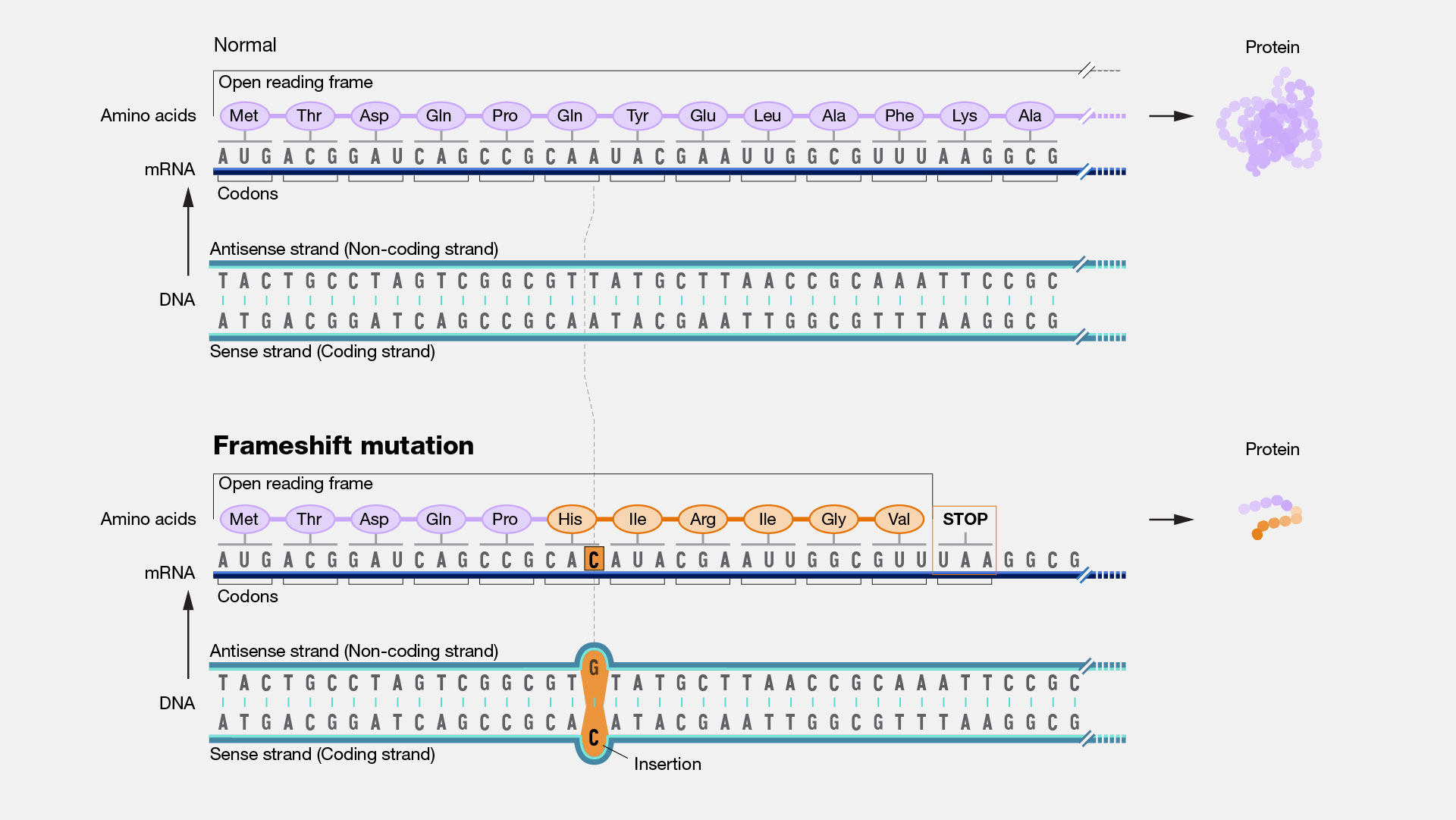
A frameshift mutation in a gene refers to the insertion or deletion of nucleotide bases in numbers that are not multiples of three. This is important because a cell reads a gene’s code in groups of three bases when making a protein. Each of these “triplet codons” corresponds to one of 20 different amino acids used to build a protein. If a mutation disrupts this normal reading frame, then the entire gene sequence following the mutation will be incorrectly read. This can result in the addition of the wrong amino acids to the protein and/or the creation of a codon that stops the protein from growing longer.
Narration
Frameshift Mutation. Imagine that you are reading a great mystery novel. When you get to an important part of the plot, you find that a chapter of the book is missing, either torn from the book or not printed correctly. Because the chapter is missing, none of the following parts of the book make much sense. For DNA, a missing segment can cause the cell to effectively stop reading from that gene. The consequences depend upon the information stored in the part of the genome where the problem occurred. If the gene performs a critical function, a significant medical problem can result.